New Publication in the HIPOBAT project
Title
Quantifying static capacity losses in solid-state battery composites via coulometric titration comparison
Link
https://doi.org/10.1039/D5CC07213A
Summary
In this work, we present a simple method called Coulometric Titration Comparison (CTC) to measure how much of the active material in solid-state battery (SSB) electrodes is actually electrochemically connected. The method compares the same cathode material in solid and liquid electrolyte systems to identify inactive mass. This is important because SSBs often show lower performance due to poor particle connectivity, not necessarily due to material limitations.
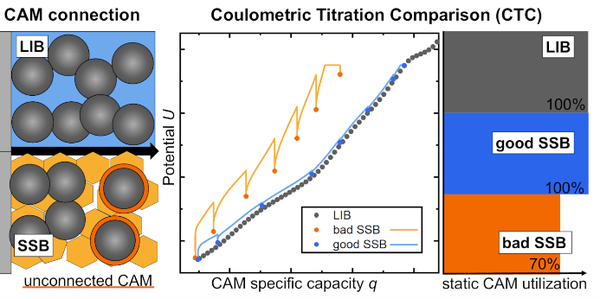
CTC allows us to quantify these so-called “static capacity losses” directly during operation. We tested the method on both well-performing (“good”) and poorly performing (“bad”) SSB electrodes. The results clearly distinguished differences in active material utilization between these cases.
To validate the approach, we compared the findings with post-mortem X-ray diffraction (XRD) measurements. The agreement between both methods confirms the reliability of CTC. Overall, this approach provides an accessible tool for better understanding and improving SSB electrode design.
Interview with the lead author and researcher in the HIPOBAT project, Kilian Vettori, Justus-Liebig-University Giessen
1. Your article describes a method recently developed in your group led by Jürgen Janek to quantify the utilization of cathode active material in solid state batteries by an electrochemical measurement called coulometric titration comparison (CTC). What was the motivation to develop this method?
The main motivation was that, in conventional cycling experiments, we measure voltage versus capacity but cannot directly distinguish between kinetic and static capacity losses. However, for solid-state batteries, it is crucial to understand how much of the cathode active material is actually electrochemically connected.
By using coulometric titration, we measure the system close to equilibrium, which allows us to largely neglect kinetic effects. This enables us to isolate and quantify the fraction of active material that is truly utilized, giving deeper insight into the quality of the cathode composite.
2. Could you please explain the difference of static and kinetic losses and how they affect the performance of electrochemical cells?
First, it is important to note that this distinction is a simplification, since capacity losses in real systems depend on many interacting factors. However, for a clear understanding, we can separate them into static and kinetic losses.
Static losses arise when parts of the active material are not electrochemically connected at all, meaning they can never contribute to the capacity. In contrast, kinetic losses occur when the material is connected, but transport limitations (e.g. slow ion or electron movement) prevent it from being fully utilized within a given time.
A helpful analogy is a parking garage with 100 cars trying to enter. In the static case, half of the parking spaces are inaccessible, for example because they are under construction. This means only 50% of the capacity can ever be used.
In the kinetic case, all parking spaces are available, but there is only one narrow entrance, so traffic moves very slowly. If we check after a fixed time, only part of the cars have entered, even though, in principle, all spaces could be filled given enough time.
3. To which materials systems have you applied the method so far? Are there any limitations. For instance, do you think that CTC can yield useful results for hybrid electrolyte systems consisting of a mixture of polymers and inorganic electrolytes and maybe even small amounts of liquids?
So far, we have mainly applied CTC to layered oxide materials in lithium-based systems. However, in principle, the method is not limited to a specific material class or electrolyte and could also be extended to other systems, including anodes.
That said, for CTC to provide meaningful results, several key conditions must be fulfilled. First, the voltage response of both the liquid and solid-state cells should reflect the same equilibrium behavior of the pristine active material, meaning they follow the same titration curve. In addition, the voltage must show a clear dependence on the inserted charge. Materials with very flat voltage plateaus, such as Lithium Iron Phosphate, can therefore be challenging.
Second, the measured charge should originate only from lithium insertion and extraction in the active material. Any side reactions, such as electrolyte decomposition, can distort the results and reduce the reliability of the method.
Regarding more complex systems, such as hybrid electrolytes combining polymers, inorganic components, and small amounts of liquid, CTC should still be applicable in principle. However, these systems may introduce additional side reactions or non-equilibrium effects, which need to be carefully controlled and validated.
4. In the HIPOBAT project we aim at developing a high-power sodium solid state battery. Have you planned to apply this method to sodium-based systems in the near future?
Definitely, yes! Currently the stability between solid electrolytes and sodium-based cathode materials is an issue, due to the mentioned reasons. But once this challenge is overcome, we plan to apply CTC to quantify utilization in sodium solid state batteries.
5. If other researchers want to perform CTC measurements, how would you recommend to start? Are there any limitations in terms of cell set-up, experimental parameters, cathode composition, etc.?
This is actually one of the strengths of CTC. It is straightforward to implement. If a researcher wants to quantify static losses in a solid-state battery electrode with a given active material mass, the starting point is to perform a coulometric titration measurement.
This involves applying small charge or discharge steps at low current, followed by relaxation periods to reach near-equilibrium conditions. In this way, equilibrium voltages can be recorded as a function of the inserted charge. Already at this stage, it becomes possible to qualitatively distinguish between kinetic and static effects.
For the full CTC analysis, the same measurement must then be repeated in a reference system where the active material utilization is well understood, e.g. in a liquid electrolyte cell.
The two resulting voltage–charge relationships can then be directly compared with the python script provided with our publication. From this comparison, the utilization of the active material in the solid-state system relative to the reference can be quantified.
In terms of limitations, it is important to ensure near-equilibrium conditions, neglectable contribution from the anode, minimize side reactions, and choose suitable materials with a well-defined voltage response. The cell design itself is relatively flexible, but the measurements must be carefully controlled to obtain reliable results.

